The 29th session of the Codex Alimentarius Committee on Fats and Oils (CCFO29) addressed all the topics on its agenda. CCFO29 main discussions focused on options to include nutritional restrictions to limit the presence of industrially produced trans-fatty acids (iTFAs) in foods covered by three Codex standard (edible fats and oils not covered by individual standards (CXS 19); named animal fats (CXS 211); and, fat spreads and blended spreads (CXS 256)). Discussions revolved around definitions and the essential composition provisions of the relevant commodities, as well as methods of analysis for iTFAs. No agreement was reached on which text to be included on these three standards, but significant progress was made on the various concepts (iTFA, total TFA, TFA naturally present in food). An intersessional work will advance discussions on how CCFO30 may agree on texts to limit iTFAs and/or prohibiting partially hydrogenated oils (PHOs). CCFO29 agreed to advance the proposed draft standard for microbial omega-3 oils for second reading process and to request the Codex Committee on Food Additives (CCFA) to discuss in which food category microbial oils may fall and, if warranted, to consider creating a separate food category for microbial oils. With regards to ongoing discussions on parameters in extra virgin olive oil related to Pyropheophytin “a” (PPP) and 1,2-diglycerides (1,2-DAGs), CCFO29 requested FAO to start an analysis of all information and data collected by the Committee so far. With regards to 4α-desmethylsterols in olive oil and olive pomace oil (CXS 33) and consistent with already adopted decision-tree, CCFO29 agreed to develop a framework for further data collection on all relevant parameters for possible revision of the standard to focus on progress on scientific data and information collection. With regards to the constant evolution of the list of acceptable previous cargoes (LOAPC) included in the Appendix II of CXC 36, CCFO29 clarified that (a) two types of mineral oils (high and medium viscosity and medium and low viscosity) should be “highly refined food-grade” (without defining what that means); retained nitric acid on the list of banned immediate previous cargoes (due to lack of data to support moving it to the LOAPC); (b) retained ethanol entry in the LOAPC without introducing any new explanatory text (about denatured ethanol); and (c) amended the template for submission of the information required for new proposals to amend the LOAPC. The amended LOAPC was sent for final adoption by CAC49. CCFO29 agreed to start another set of revisions to the Standard for named vegetable oils (CXS 210), with the aim to include shea butter; sea buckthorn oil; and, to adjust the stearic acid content in high-oleic-acid sunflower-seed oil.1
See more information about the references quoted in this article available freely in endnotes (2) to (4) respectively on CCFO29 working documents2, the official report of the CCFO29 meeting3, and Codex texts developed by the Committee (standards, guidelines, codes of hygienic practices, and related miscellaneous texts, and statements).4
NO CONSENSUS FOUND ON HOW TO REGULATE TRANS FATTY ACIDS IN THREE EXISTING CODEX STANDARDS TO HELP REDUCING THE DIETARY INTAKE TO TRANS FATTY ACIDs (TFA), FOCUSING ON THOSE TFA RESULTING FROM SOME FOOD MANUFACTURING PROCESSES (e.g., iTFA IN SOME VEGETABLE OIL HYDROGENATION PROCESSES)
General context
WHO recalled that it started the work for a global elimination of iTFA in food in 2018, and by end 2025, 65 countries had adopted best-practice policies (six countries having adopted such policies since 2024) and that about half of the global population remained unprotected from the dietary exposure to iTFA. WHO continued support to Member States through technical assistance, policy guidance, capacity-building workshops, and practical tools. In particular, WHO launched a “TFA validation program” in 2023, to recognize countries that have implemented best-practice iTFA policies supported by effective monitoring and enforcement. Validation certificates were awarded to nine countries in 2024 and 2025.
WHO also recently issued a call for global data on fatty acid composition of bovine milk to support validation of iTFA (and TFA) dietary intake estimation approaches. WHO reported also on the recent publications of new WHO guidelines on healthy diets and policy actions (especially healthy school environments), as well as about those under development.
Main outcome
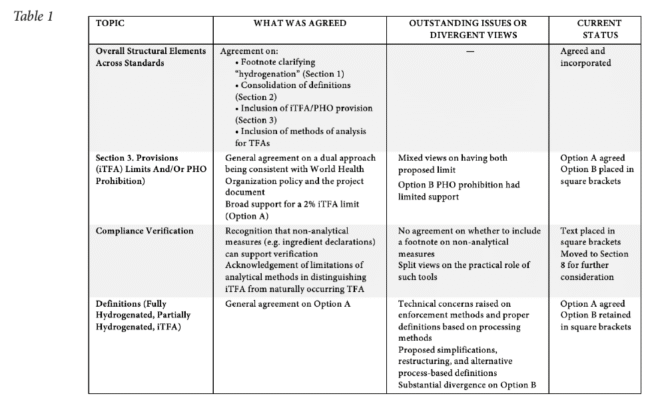
CCFO29 considered the outcome of the work on an in-session working group (IWG), itself considering the outcome of an intersessional electronic working group (EWG), both led by Canada and co-led by Saudi Arabia, suggesting changes to three Codex standards covering edible fats and oils not covered by individual standards (CXS 19), named animal fats (CXS 211), and fat spreads and blended spreads (CXS 256). CCFO29 noted progress made on the definitions and associated notes on full and partial hydrogenation processes, the proposal to introduce a maximum limit of 2 g iTFA per 100 grams total fatty acids (2%) for all industrially produced trans fatty acids (iTFA), and remaining points on validation and certification and methods of analysis, for further discussions (such as the partial hydrogenation link to the generation of iTFA).
Due to lack of general consensus on the terms and definitions (e.g., how drawing a line on what would constitute a partial hydrogenation of vegetable oil which may lead to iTFA presence or not) and how best incorporating changes to the three standards at stake (e.g., a maximum limit for the presence of iTFA; or a ban of using PHOs; or both), CCFO29 agreed to return the proposed draft revisions for a complete redrafting. For that purpose, CCFO29 established an EWG, under the lead of Saudi Arabia and co-lead of the European Union to revise both the definitions and the essential composition provisions and related aspects on checking for compliance (i.e., the methods of analysis), taking into consideration all the comments submitted to and discussions held at CCFO29, including the changes made by the CCFO29 session as contained in CRD38. The possibility of holding any intersessional physical and/or virtual working group(s) in advance of CCFO30 to consider the proposed draft revisions was kept opened. The outcome of the EWG report will be considered in advance to and at the CCFO30 meeting (March 2028).5
Main elements discussed (progress made)
Scope: existing reference to hydrogenation – footnote removed but concept kept in scope for future discussions
CCFO29 considered the proposed footnote intended to clarify the term “hydrogenation”, and noted the following views: (a) the footnote provided no added value and should be deleted; (b) the footnote was not appropriate for inclusion under the scope as it could give the impression that partially hydrogenated fats and oils were within the scope of the respective standards; (c) the footnote provided no clear distinction between PHO and Fully Hydrogenated Fats and Oils and should not be included in the scope to avoid misinterpretation; and (d), the word “full” could be included before hydrogenation to improve clarity; however, concerns were expressed with this approach as it was technically inaccurate. CCFO29 therefore agreed to remove the proposed footnote and maintain the word “hydrogenation” in the scope of CXS 19, but without any further clarification (i.e., status quo with no change to the scope of the current (2024) version of published CXS 19).
Product definitions: Fully Hydrogenated Fats and Oils / Full hydrogenation of fats and oils
CCFO29 considered product-focused and process-focused definitions, and noted concerns raised regarding (a) the clarity of the definitions, that were geared towards supporting the methods for identification and control of hydrogenated products; (b) the lack of content in the definition to confirm or differentiate “full hydrogenation” from “partial hydrogenation”; (c) the importance of indicating the transformation processes that produce trans-fatty acids (TFA) within the definition; and (d) the complexity of the definition, noting that some of the technical terms therein (e.g. isomerization; cis configuration) might require further definition to aid understanding.
With the removal of the footnote to “hydrogenation” in the scope of the standard CXS 19, which was the only place that the term “full hydrogenation” appeared in the standard, the definition of “fully hydrogenated fats and oils” may no longer be necessary. Recalling that definitions should only be provided to facilitate the understanding of the content of the standard, CCFO29 agreed to delete the proposed definition for Fully Hydrogenated Fats and Oils and for Full hydrogenation of fats and oils.
The two unagreed – and now fully deleted – proposed definitions did read as “[Fully hydrogenated fats and oils (FHO) are produced through a chemical process in which hydrogen is added to all the double bonds in unsaturated fats and oils, using a catalyst. This process converts all unsaturated bonds into saturated single bonds, resulting in a fully saturated fat structure. Because the hydrogenation is complete, the formation of industrially produced trans-fatty acids (iTFA) is minimized. The resulting fats and oils are typically solid or semi-solid at room temperature due to their higher melting point and exhibit enhanced stability and resistance to oxidation.] / [Full hydrogenation of fats and oils is an industrial chemical process in which through the addition of hydrogen, all unsaturated bonds are converted to saturated single bonds resulting in a fully saturated fat structure. Because the hydrogenation is complete, the formation of industrially produced trans-fatty acids (iTFA) is minimized [to less than
2%]. The resulting fully hydrogenated fats and oils are typically solid or semi-solid at room temperature due to their higher melting point and exhibit enhanced stability and resistance to oxidation.]). Members were of the view that the proposed definitions were overly long and complex and did not need a complete description of the partial hydrogenation process; they contained unfamiliar chemical terminology which may need further explanation; ranges or thresholds for iTFA to make the connection between PHO and iTFA should be included; iTFA cut-off or iTFA threshold should not be included since iTFA could originate from other processes; and information was lacking to support analytical-based approaches in enforcement. Proposals to include a range or value for iTFA (“25-45% iTFA”; or “more than 2% iTFA”) were not supported.
The proposed unagreed definition – subject to redrafting – for industrially produced trans-fatty acids (iTFA) would read as being those “[trans-fatty acids formed through industrial processes, [primarily during the partial hydrogenation of unsaturated fats and oils, though smaller amounts may also be produced during other industrial processes, such as oil refining and deodorization]. [iTFA are defined solely by their method of production and are difficult to be chemically distinguished from [other naturally occurring ruminant] TFA.].]. Members expressed a range of views such as: examples of refining processes which are generating small quantities of iTFA should not be included; all processes that contributed to the formation of iTFA should be included; the last sentence should be deleted as it went beyond the scope of a definition on iTFA; the term “other TFA” should be replaced with “naturally occurring TFA” in the last sentence of the definition; the definition should not include a reference to methodology; to explain methodological challenges and difficulties in analytically distinguishing iTFA from other TFA in the definition was not warranted.
Section 3: Essential composition and quality factors
CCFO29 discussed a dual approach to reducing dietary intake of industrially produced trans fatty acids (iTFA): Option A, a numerical limit of 2 g iTFA per 100 g total fatty acids, and Option B, a prohibition of all partially hydrogenated oils (PHO). While the WHO confirmed that both approaches are internationally recognized best-practice measures under the REPLACE initiative, Members were divided on whether both should be included in a Codex standard.
Two clear positions emerged. Some Members supported Option A only, arguing that Codex standards should focus on objective, measurable product specifications to avoid trade disruptions and enforcement challenges. They emphasized that the 2% iTFA limit is verifiable, whereas PHO prohibitions are difficult to monitor due to the lack of universally accepted analytical methods and are better addressed through national legislation. Other Members supported retaining both Options A and B, stressing the need for flexibility to accommodate diverse national regulatory frameworks and WHO policy guidance. They noted that PHO prohibitions are an effective upstream measure, particularly for countries with limited laboratory capacity, and that prohibitions already exist in some Codex texts.
Despite extensive discussion, CCFO29 remained divided. An informal drafting process proposed revised text including: (i) an introductory clause linking application of the provisions to national and regional legislation; (ii) retention of the 2% iTFA limit; and (iii) a requirement that products “shall not be partially hydrogenated,” reframed as a compositional characteristic. While some Members viewed this as a flexible compromise, others raised concerns about enforceability, divergent national application, and potential trade barriers.
No agreement was reached on how to reflect both approaches within the standard, and progress was further constrained by the lack of consensus on key definitions, including “partial hydrogenation” and “industrially produced trans fatty acids.” As a result, CCFO29 agreed that further discussion would be required, and the issue revisited at CCFO30.
PROPOSED DRAFT STANDARD FOR MICROBIAL OMEGA-3 OILS – CONSENSUS FOUND TO MOVE THE REVISED TEXT TO THE NEXT ELABORATION AND READING PHASE
Main outcome
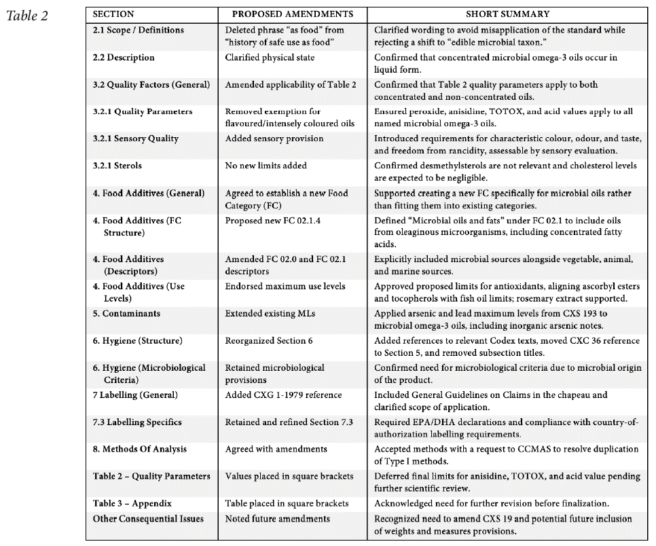
CCFO29 agreed to send the revised proposed draft standard for microbial omega-3 oils to CAC49 for to advance it into the development process. CCFO29 agreed accordingly to forward the provisions for labelling and methods of analysis to CCFL and CCMAS respectively for endorsement, noting that one of the methods may need to be “retyped” by CCMAS. Similarly, CCFO29 forwarded the proposed food additive provisions to CCFA for endorsement. With regards the section on contaminants, CCFO29 requested CCCF to consider extending the MLs of arsenic and lead for edible fats and oils in CXS 193, as well as the notes and remark associated with the ML for arsenic to microbial omega-3 oils. CCFO29 sought CCFA advice on the appropriate food category to use in CXS 192, where products conforming to the future standard on microbial omega-3 oils would fall, noting that CCFO was unable to assign a food category for microbial omega-3 oils, since according to the GSFA Food Category (FC) System, neither FC 02.0 (Fats, oils and emulsion), nor its sub-categories currently include oil from microbial sources. CCFO29 expressed its preference for establishing a new sub-category for oils of microbial origin; and, FC 02.0 and FC 02.1 would need to reflect microbial oils and the reference to triglycerides in FC 02.1 should be broadened to reflect the different types of microbial oils for human consumption.
CCFO29 agreed to establish an EWG, under the lead of the USA and co-lead of China to consider all the outstanding issues placed in square brackets in the text of the draft standard, while taking into account the discussions at CCFO29, and the comments expected to be submitted in reply to an upcoming circular letter after CAC49 decision. The EWG was also tasked to elaborate proposed draft consequential revisions to be made to the standard CXS 19, once the draft standard for microbial omega-3 oils would be finalized and adopted by the CAC. The possibility to convene a pre-session working group in advance of CCFO30 to further refine the draft standard was kept open.6
OLIVE OIL AND POMACE OLIVE OIL, INCLUDING EXTRA VIRGIN OLIVE OIL
General context
The International Olive Council (IOC) representative thanked the CCFO Chairperson and the Codex Secretariat for their ongoing support and collaboration and recalled its long-standing cooperation with Codex to harmonize standards, protect consumers, enhance product quality, facilitate trade, and prevent fraud. In addition to the work of improving the Codex standard on olive oil and pomace oil (CXS 33) engaged since 2013, IOC contributed to the alignment of the Codex standard for table olives (CXS 66) the IOC standard. The representative highlighted that IOC develops its standards based on robust scientific evidence and official data from all producing countries, supported by the engagement of over 178 experts, 44 working groups, 141 sensory panels, and 125 testing laboratories. Over the years, at Codex’s request, the IOC had also conducted significant studies, including those on fatty acids, campesterol, and delta-7-stigmastenol, and continued to advance research in areas such as PPP, DAGs, sterols, and olive pomace oils. The representative reaffirmed IOC’s commitment to support Codex by offering its scientific expertise and resources to ensure objective and transparent studies as the leading intergovernmental authority on olive oil and table olives.
Pyropheophytin “a” (PPP) and 1,2-diglycerides (1,2-DAGs) in extra virgin olive oil
CCFO29 based its discussions on the outcome of an intersessional EWG and a further proposal put forward by Italy, with the help of Australia, Canada, Saudi Arabia, and the USA, as included in CCFO29 CRD 36. CCFO29 agreed to follow a kind of hybrid path that would include (a) clearly defining the scope of the analysis to be performed by the FAO; (b) transmitting all the available data and information to FAO; and (c) requesting FAO to use the same data collection template and criteria previously issued in CL 2024/36-FO, so that all new data submissions remained comparable with data previously collected by the EWG. Noting the concerns expressed about the timelines, it was proposed to request FAO to give priority to this request and report to CCFO30 (in 2028). See final CCFO29 report for more details.
CCFO29 hence agreed to request FAO to convene an expert consultation to review and analyze the information submitted to the EWG in response to CL 2024/36-FO, as well as any additional data related to the completion of ongoing studies on the evolution of pyropheophytin “a” (PPP) and 1,2-diglycerides (1,2-DAGs) in extra virgin olive oil (EVOO) and information complying with the Annex to CL2024/36-FO, within the timeframe established by FAO in the related call for data and provide advice on: (a) whether the observed values for PPP and 1,2-DAG are systematic, reproducible, and correlated to the other quality parameters listed in annex of CL 2024/36-FO; (b) whether PPP and 1,2 DAG’s could be used as quality parameters of EVOO along with other quality parameters including sensory in CXS 33-1981; and (c) the overall robustness and discriminatory power of PPPs and 1,2-DAGs as quality parameters. Additional data and information may continue to be collected and submitted directly to FAO in response to its future call for data (in advance to an expert consultation) in using the same data collection template and criteria previously issued in CL 2024/36-FO.
4α-desmethylsterols in olive oil and olive pomace oil (CXS 33)
Given the complexity of this issue (technically, analytically, economically, etc.), CCFO29 agreed to develop a framework for further data collection on all relevant parameters for possible revision of the standard to focus on progress on scientific data and information collection, consistent with already adopted decision-tree. See final CCFO29 report for more details.
CCFO29 agreed to establish an EWG, under the leadership of the Syrian Arab Republic, with the help of Australia, Saudi Arabia, Tunisia, and the IOC to (i) define the types of data to be collected, including sterol composition and relevant quality parameters; (ii) identify analytical methods and sampling approaches, drawing on internationally recognized methods; (iii) propose a targeted timeline for data collection, taking into account the multi-season nature of olive production; (iv) prepare a template based on the above to be included in a CL to be issued by the Codex Secretariat to inform all Members and Observers of this initiative and request them to submit the required data; and, (v) prepare a report to be considered at CCFO30 (in 2028). CCFO29 also agreed to inform CCEXEC that the completion of the work on footnote (c) would require sufficient time for data collection and was not expected to be completed by CCFO30, so that CCFO30 would likely ask for a revised timeline.
Category of ordinary virgin olive oil (pending discussions held at CCFO27)
Regarding the category of ordinary virgin olive oil added to CXS 33, it was recalled that CCFO27 agreed to reconsider the issue at CCFO30 based on the earlier decision by CCFO26 to remove the category, in response to requests from several Members, including developing countries, who sought additional time to examine the implications of the deletion, particularly given its potential impact on trade and the economic consequences for producers. CCFO27 did agree to temporarily retain the definition of ordinary virgin olive oil, along with its associated footnotes and related parameters, until CCFO30, to allow Members and Observers sufficient time to complete the necessary scientific work to be undertaken that would support decision-making. Members were reminded to fulfill their CCFO27 commitment and undertake the required work ahead of CCFO30, and they were encouraged to attend CCFO30 well prepared, to allow well informed and efficient discussion and possibly reach a final decision on that matter.
REVIEW OF THE LIST OF ACCEPTABLE PREVIOUS CARGOES (APPENDIX II TO CXC 36) “LOAPC” (no MOAH ML in type II and III, add “highly refined food-grade” qualifier to other mineral oil types, ethanol/denatured ethanol, nitric acid)
CCFO29 discussed four main recommendations resulting from the EWG report: (a) proposal to limit MOAH at 2.0 mg/kg in (i) mineral oil, medium and low viscosity, class II (highly refined food-grade) and (ii) mineral oil, medium and low viscosity, class III (highly refined food-grade); (b) proposal to maintain miner- al oil, high viscosity (CAS number 8012-95-1) and mineral oil, medium viscosity in the List of Acceptable Previous Cargoes, but to add a reference as “highly refined food-grade” to both of them (consistent with previous CCFO decision on the ones mentioned in (a)); (c) recommendation to remove nitric acid (CAS number 7697-37-2) from the List of Banned Immediate Previous Cargoes and its inclusion in the List of Acceptable Previous Cargoes; (d) recommendation to amend the entry of ethanol (CAS number 64-17-5) to clarify the status of denatured alcohol; (e) to develop of a standard template table for submission of information required for proposals to amend the List of Acceptable Previous Cargoes.
CCFO29 agreed that a limit of 2.0 mg/kg MOAH for highly refined food-grade mineral oil, medium and low viscosity, class II and mineral oil, medium and low viscosity, class III should not be established, opting to retain the substances as previously listed in the LOAPC.
CCFO29 endorsed the EWG’s recommendation to retain mineral oil, high viscosity and mineral oil, medium viscosity in the LOAPC, and agreed to amend both entries by adding the wording “highly refined food-grade” to these substances, to read “Mineral oil, high viscosity (highly refined food-grade); Mineral oil, medium viscosity (highly refined food-grade)”. The European Union expressed its reservation regarding the retention of mineral oil high viscosity (CAS number 8012-95-1) and mineral oil medium viscosity in Appendix 2 of CXC 36, citing concerns that the MOAH fraction of these mineral oils could contain certain genotoxic carcinogens and would present potential health risks associated with consumer exposure.
CCFO29 agreed that nitric acid (CAS number 7697- 37-2) would remain on the list of banned immediate previous cargoes (LBIPC) and to reconsider it only when adequate supporting information is provided in the future.
CCFO29 agreed to retain the original ethanol entry in the LOAPC without introducing any new explanatory text and noted that future consideration of denaturants used in ethanol would require submission of data for JECFA evaluation, after which the matter could be revisited. The EU expressed its reservation to the decision to not include the qualifying text proposed by the EWG because ethanol is also even often transported in a denatured form. Denatured ethanol should only be acceptable as a previous cargo, provided that the denaturant was also included in the LOAPC, which meant that it had been subject to risk assessment. It was not appropriate to only require that the denaturant to not be included in the list of banned substances, as proposed in the EWG initially, since this would allow the presence of all possible denaturants, which have not yet been assessed. There were alternative denaturants already in the LOAPC, and therefore the proposed qualifying text was reasonable.
In summary, CCFO29 agreed to forward the draft amendment to the List of Acceptable Previous Cargoes (i.e., Appendix II of CXC 36), to the CAC49 for adoption, as included in Appendix IV – Part 1 of the report of the CCFO29 with the two changes to the entries on mineral oil with high viscosity (CAS RN 8012-95-1) and with medium viscosity with the added qualifier “highly refined food-grade” and requested the Codex Secretariat to issue a new circular letter, including the draft amended template table based on CCFO29 discussions, as included in Appendix IV Part 2 to the CCFO29 report, and to invite interested Members and Observers to (a) propose further amendments to Appendix 2: List of Acceptable Previous Cargoes of CXC 36; and (b) provide further comments on the draft template table for submission of future proposals for amendments to that LOAPC.
CCFO29 also agreed to establish a new EWG, under the lead of Malaysia to (a) consider proposals on new substances to be added to the LOAPC, provided that such proposals are supported by adequate and relevant information; (b) prioritize substances, if any, to be submitted to JECFA for evaluation; (c) consider proposals to remove substances from the list in light of new data; (d) review the proposed draft template to be used as a standard template for proposals to amend the List of Acceptable Cargoes; and (e) prepare a report for consideration by CCFO30. The possibility of holding intersessional PWG/VWG(s) in advance of CCFO30 to advance that work was also kept open.7
METHOD OF ANALYSIS FOR THE DETERMINATION OF GAMMA ORYZANOL IN RICE BRAN OIL
CCFO29 recognized that the method for the determination of gamma oryzanol in CXS 234 continued to be ft for purpose. CCFO29 agreed to forward the method and its characterization (Provision, Method, Principle, and Type) to CCMAS for re-endorsement; retyping as Type III (considering the interlaboratory studies carried out in one country) and subsequent inclusion in CXS 234 (as included in the Appendix II of CCFO29 report). CCFO 29 noted the ongoing ISO efforts to adopt two validated alternative methods for the determination of gamma oryzanol. CCFO29 encouraged Codex Members and Observers to provide feedback to both CCFO and CCMAS, once such methods would be finalized and published, for their potential inclusion in CXS 234.8
OTHER ISSUES AND FUTURE WORK
Proposed inclusion of shea butter derived from the nuts of the shea tree (Vitellaria paradoxa, formerly Butyrospermum parkii) in the standard for named vegetable oils (CXS 210)
The work will include defining the requirements for both refined and unrefined shea butter. Currently, there is an existing Regional standard for unrefined shea butter (Africa) (CXS 325R) whose scope is limited to unrefined product and it is considered as a “regional agreement” which has limited influence on other regions. A revision to CXS 210 to include shea butter would enable Codex Member countries and the food industry to appropriately characterize, name, and market shea butter, into both refined and unrefined categories, globally. The current regional standard CXS 325R specifies the quality requirements for unrefined shea butter, however the use of refined shea butter in the food industry is on the rise and therefore needs further specification. The refining process removes the scent and color of the product which makes the fat more suitable for food applications where a bland taste and clear color is required.9
Revision of the fatty acid (stearic acid C18:0) in the specifications for high oleic acid sunflower seed oil in the standard for named vegetable oils (CXS 210) “to facilitate its commercialization conditions”
High oleic acid sunflower seed oil is one of the most suitable oils for the formulation of frying oils according to the latest recommendations of the World Health Organization. Low saturation and high heat resistance due to its oleic fatty acid content are unique features of this oil, which are mainly cultivated in Eastern Europe (such as Ukraine), and its demand is increasing day by day due to its higher heat-stable quality. The main aspects to be covered is an amendment of the stearic acid content of high oleic acid sunflower seed oil, in CXS 210, such that the acceptable range is amended to lower the current lower bound of content range from “2.9% – 6.2%” to “2.1% – 6.2%”.10
Adding requirements for seabuckthorn pulp oil and seabuckthorn seed oil derived from the berry pulp and seeds: (i) Seabuckthorn pulp oil obtained from berry pulp of Hippophae rhamnoides; and, (ii) Seabuckthorn seed oil obtained from seeds of Hippophae rhamnoides, in the standard for named vegetable oils (CXS 210)
CCFO29 agreed to submit the project document on the revision to CXS 210 to include shea butter to CAC49 for approval and inform CCAFRICA of the proposed new work and that it may potentially be requested to review the need for CXS 325R once the work is completed. CCFO29 established a EWG under the leadership of Nigeria, with the support of Ghana, India, and the Netherlands to prepare proposed draft revisions and consideration by CCFO30.11
Other matters
With regards to CCFA’s request to confirm technological justifications for use of lauric alginate ethyl ester (INS 243) as a preservative (antimicrobial agent) in products conforming to the Codex standard for fat spreads and blended spreads (CXS 256), CCFO29 couldn’t reach a consensus. Some countries reported that it was permitted for that use to prevent the growth of spoilage microorganisms, including bacteria, yeasts, and molds in products such as margarine and margarine-like table spreads, mayonnaise and spoon-able and pour-able salad dressings, at use levels up to 200 ppm. Its efficacy was attributed to its stability in the aqueous phase, which was the one commonly supporting such microbial proliferation. Other countries were not aware of such a technological effect, and it was noted that sorbates (INS 200, 202-203) and benzoates (INS 210-213) were the only preservatives with a numerical acceptable daily intake (ADI) permitted for use in some types of fat spreads and blended spreads conforming to that Standard. CCFA56 was further invited to consider that state of play.
The publication is posted in the February 2026 World Food Regulation Review: https://cloud.3dissue.com/170388/199109/233437/WFRR-Vol35nr09-Feb2026/index.html. Find the full WFRR library here.
ENDNOTES
- The Codex Alimentarius Committee on Fats and Oils (CCFO) is the specialised body which elaborates standards for fats and oils of animal, vegetable and marine origin, including margarine and olive oil. CCFO29 meeting was held in Kuala Lumpur (Malaysia) from 9 to 13 February 2026. CCFO29 was chaired for the first time by Ms Zailina Abdul MAJID, Deputy Director-General of Health (Food Safety and Quality) from the Ministry of Health of Malaysia. CCFO29 was attended by about 150 delegates from 50 Member Countries, one Member Organization (European Union) and 7 Observer organizations and FAO and WHO. It was also attended by two International Governmental Organizations (i.e., the Inter-American Institute for Cooperation on Agriculture (IICA) and the International Olive Council (OIC)). Subject to confirmation, the next session of the Committee (CCFO30) was tentatively scheduled to be held in two-year time, from 20 to 24 March 2028. See also Codex website news page about CCFO29 outcome at https://www.fao.org/fao-who-co-dexalimentarius/news-and-events/en/. ↩︎
- See https://www.fao.org/fao-who-codexalimentarius/ meetings/detail/en/?meeting=CCFO&session=29& ↩︎
- See https://www.fao.org/fao-who-codexalimentarius/ meetings/en/ ↩︎
- See https://www.fao.org/fao-who-codexalimentarius/ committees/committee/related-standards/en/?commitee=CCFO ↩︎
- CRD38 (fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fcodex%252FMeetings%252FCX-709-29%252FCRDs%252Ffo29_crd38x.pdf
Canada, as Chair of the EWG and IWG, speaking also on behalf of Saudi Arabia, as Co-Chair, summarized the work undertaken by the EWG and the discussions held in the IWG to revise the Standards for edible fats and oils not covered by individual standards (CXS 19), named animal fats (CXS 211), and fat spreads and blended spreads (CXS 256) to support the WHO REPLACE initiative aimed at reducing the intake of industrially produced trans-fatty acids (iTFA). Here below is a synthetic summary of main outcome and outstanding issues that CCFO29 discussed. See Table 1 below.
Despite these unresolved elements, it was considered that the EWG and IWG had made substantial progress, producing a structured draft and a set of identified issues for plenary discussions. The importance of preserving flexibility in the standard, reflecting the two WHO endorsed regulatory pathways, was highlighted. CCFO29 plenary attempted to make further progress on these three elements, especially on the definitions, based on the report presented in CCFO29 CRD02 (see https:// www.fao.org/fao-who-codexalimentarius/sh-proxy/en/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FMeetings%252F-CX-709-29%252FCRDs%252Ffo29_crd02x.pdf).
↩︎ - See the text of the draft standard in the Appendix III of the CCFO29 report. CCFO29 considered the outcome of an intersessional EWG, under the lead of the USA with the help of China as well as a revised version of the text resulting from additional comments submitted in response to the circular letter on the report of the EWG (CL 2025/80-FO). CCFO29 therefore based its discussions on that text presented in CCFO29 CRD21. The proposed amendments are summarized as shown in Table 2 below. ↩︎
- See Appendix IV, part 1 and part 2 of CCFO29 report. ↩︎
- See Appendix II of CCFO29 report. CCFO29 recalled that, at its previous session, discussions regarding the transfer of the method for determining gamma oryzanol in rice bran oil transcribed in the Standard for named vegetable oils (CXS 210) were deferred to this session. The Codex Secretariat presented a summary of the responses to the CL and these generally indicated:
(i) support for transferring the current UV spectrophotometric method from CXS 210 to CXS 234 as Type IV; (ii) confirmation of the method as fit for purpose; and (iii) ongoing work to develop alternative methods by the International Organisation for Standardisation (ISO), among others. CCFO29 noted the following comments and views concerning the method: (a) Fitness for purpose: based on analytical experience, the method was easy to use; simple and accessible by laboratories due to its low cost and it was therefore ft for purpose; (b) Interlaboratory testing: the method had been assessed through inter-laboratory tests across twelve laboratories and the results confirmed that the method was efective; (c) Alternative methods: two alternative methods for the determination of gamma oryzanol in rice bran oil (spectrophotometer method and high-per- formance liquid chromatography method) were under development within ISO, that should these two standards be published, they should also be recommended to CCMAS for their inclusion in CXS 234. ↩︎ - See Appendix V of CCFO29 report. ↩︎
- See Appendix VI of CCFO29 report. ↩︎
- See Appendix VII of CCFO29 report. ↩︎